A Positive Feedback loop for Gene Silencing
Max Planck Researchers decipher the role of a long-known histone modification and thereby uncover an unexpected mechanism for silencing genes
The packaging of DNA by histone proteins into chromatin regulates many processes, including the decision of whether genes are transcribed or whether they are silent. The addition of specific chemical tags on histone proteins mark genes as being silenced. One of these tags is the covalent linkage of a small protein called ubiquitin to one of the histones. This modification was discovered more than 30 years ago but its molecular function has remained elusive. Scientists of the Max Planck Institute of Biochemistry (MPIB) in Martinsried near Munich, Germany now identified a protein complex that binds this ubiquitin modification and show how this is connected to gene silencing.
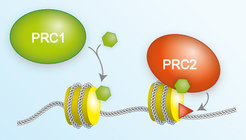
The Polycomb Repressive Complex 1 (PRC1) modifies nucleosomes - a complex made of histone proteins (yellow cylinder) and DNA (white double helix) - by linking a small molecule called ubiquitin (green hexamer) to a specific site on one of the histone proteins. The ubiquitin tag promotes binding of Polycomb Repressive Complex 2 (PRC2) that in turn then adds a methylation tag (red triangle) to a specific site on one of the other histone proteins.
While all cells in the human body carry the same genetic information within their DNA, they significantly differ in their form and function. This is achieved via a cell type-specific gene regulation, in which genes can be either switched ‘on’ or ‘off’. How this is mediated at the molecular level is not fully understood. However, modifications of the histone proteins that package DNA into chromatin are known to play a crucial role.
One of these modifications was discovered more than 30 years ago: the monoubiquitinated form of histone H2A. Ten years ago, an important step towards understanding the role of H2A monoubiquitination was made through the discovery of the enzyme that adds the ubiquitin tag on H2A - the Polycomb repressive complex 1 (PRC1). This protein assembly is required for the long-term silencing of genes. However, the molecular mechanism by which the ubiquitin tag on H2A brings about gene silencing still remained unknown.
“To find out what the ubiquitin tag on H2A does, we teamed up with our collaborators and searched for proteins that bind this tag and thereby identified another Polycomb protein complex”, explains Reinhard Kalb, scientist at the MPIB. “This complex, called PRC2 turned out to be the main interacting factor that binds the ubiquitin tag on H2A in both fly and mouse cells.” PRC2 itself modifies another histone, and this modification in turn recruits PRC1. “We propose that, together, these enzymes create a feed-back loop which contributes to gene silencing”, summarizes Jürg Müller and adds, “the next step is now to find out how this positive feedback loop functions in vivo to set up Polycomb-silenced chromatin.”
[HS]
Original Publication:
R. Kalb, S. Latwiel, H. I. Baymaz, P. W. Jansen, C. W. Müller, M. Vermeulen and J. Müller: Histone H2A monoubiquitination promotes histone H3 methylation in Polycomb repression. NSMB, May 18, 2014.
DOI:10.1038/nsmb.2833






