Protein synthesis in the power plants of the cell
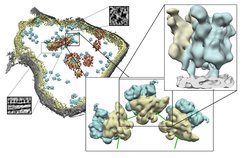
Three-dimensional structure of a mitochondrion with increasing level of details
Left: Surface representation of a mitochondrion showing the outer (grey) and the inner (yellow) membrane, mitoribosomes (blue) as well as further protein complexes (red). The small boxes show examples of the original pictures. Center: Typical assembly of mitoribosomes forming a polysome. Large (blue) and small (yellow) subunits are shown. Right: Structure of a mitoribosome (yellow and blue) bound to the inner membrane of the intact mitochondrion.
All biochemical processes in a cell require energy, which is provided by the mitochondria in form of the carrier molecule ATP. In order to produce ATP, the mitochondria require certain proteins themselves. But where do these proteins originate from? The majority is imported from the cell plasma into the mitochondria. Their building plans are stored in the genetic substance located in the nucleus, the DNA. However, a small number of the proteins necessary for the generation of energy are produced right on-site by the so-called mito-ribosomes. While the architecture of the ribosomes localized in the cell plasma has been examined intensely during the last years, scientists have only recently started to investigate the structure of the mito-ribosomes.
For the first time now, researchers in the group „Modeling of Protein Complexes“ have managed to unravel the mito-ribosome structure in its natural environment at the inner mitochondrial membrane. Here, the mito-ribosomes assemble into large complexes called polysomes. In this way, they jointly produce many copies of the same protein together. Moreover, the scientists were able to demonstrate how the association of the ribosomes with the mitochondrial surface is mediated. “We believe that the membrane protein Mba1 is a receptor for the ribosome and beyond that supports the integration of newly synthesized proteins into the mitochondrial envelope”, speculates Friedrich Förster, head of the research group.
Such detailed insights into the architecture and contact points of the mito-ribosomes are made possible by a technology named cryo-electron tomography: in a high-speed freezing process the cells are cooled down to minus 170°C, hereby maintaining the natural structure and position of the ribosomes inside the cell. In a consecutive step, the researchers take a series of pictures from many different angles, which then are computationally merged into a three-dimensional image.
Original publication
Pfeffer, S., Woellhaf, M.W., Herrmann, J.M., Förster, F.: Organization of the mitochondrial translation machinery studied in situ by cryo-electron tomography. Nature Commun, January 22, 2015
Doi: 10.1038/ncomms7019
