Wolfgang Baumeister
Emeritus Group Molecular Structural Biology
Cryo-Electron Tomography, Electron Microscopical Structure Research, Protein and Cell Structure, Protein Degradation
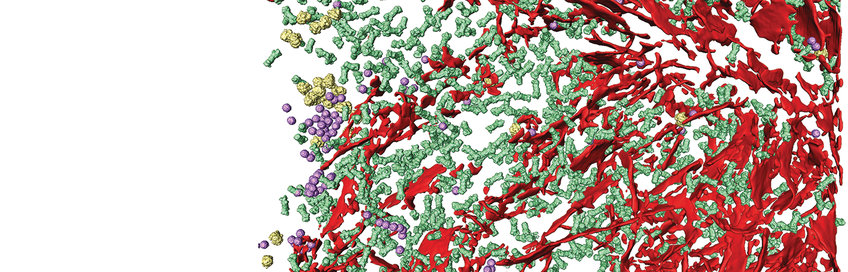
Large protein complexes, such as the 26S proteasome, perform important tasks in living cells. However, they are often very unstable or only form for a short time and can therefore hardly be detected in biochemical preparations and purified form. For this purpose, Wolfgang Baumeister's Department “Molecular Structural Biology” has developed the cryo-electron tomography. This method enables the researchers to visualize the macromolecular structures in a functional and true-to-life state in shock-frozen cells. Images from different projection angles are recorded under the microscope and mathematically combined into a 3D image cube (the tomogram). The researchers of the department use this method to investigate macromolecular complexes of prokaryotes and cell components of eukaryotes.
